Punjab State Board PSEB 12th Class Chemistry Book Solutions Chapter 11 Alcohols, Phenols, and Ethers Textbook Exercise Questions and Answers.
PSEB Solutions for Class 12 Chemistry Chapter 11 Alcohols, Phenols, and Ethers
PSEB 12th Class Chemistry Guide Alcohols, Phenols and Ethers Intext Questions and Answers
Question 1.
Write IUPAC names of the following compounds:
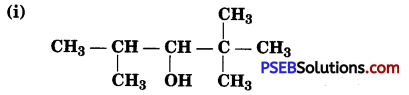
Answer:
(i) 2, 2, 4-Trimethylpentan-3-ol
(ii) 5-Ethylheptane-2,4-diol
(iii) Butane-2, 3-diol
(iv) Propane-1, 2, 3-triol
(v) 2-Methylphenol
(vi) 4-Methylphenol
(vii) 2, 5-Dimethylphenol
(viii) 2, 6-Dimethylphenol
(ix) l-Methoxy-2-methylpropane
(x) Ethoxybenzene
(xi) 1-Phenoxyheptane
(xii) 2-Ethoxybutane
Question 2.
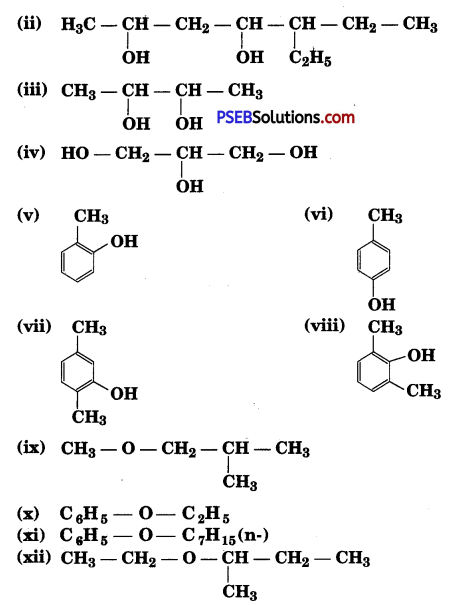
Write structures of the compounds whose IUPAC names are as follows:
(i) 2-Methylbutan-2-ol
(ii) l-Phenylpropan-2-ol
(iii) 3,5-Dimethylhexane -1, 3, 5-triol
(iv) 2,3 – Diethylphenol
(v) 1 -Ethoxypropane
(vi) 2-Ethoxy-3-methylpentane
(vii) Cyclohexylmethanol (viii) 3-Cyclohexylpentan-3-ol
(ix) Cyclopent-3-en-l-ol
(x) 3-Chloromethylpentan-l-ol.
Answer:
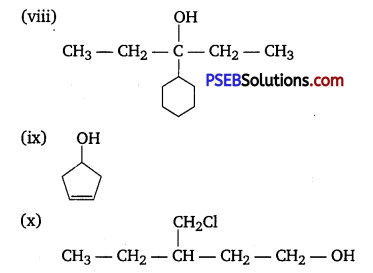
Question 3.
(i) Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
(ii) Classify the isomers of alcohols in question 3(i) as primary, secondary, and tertiary alcohols.
Answer:
(i) Eight isomers are possible. These are :
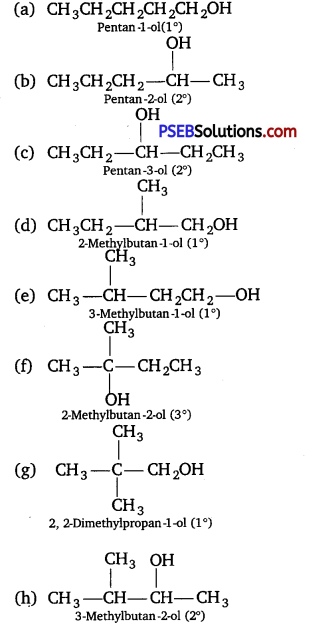
(ii) Primary : (a), (d), (e) arid (g); Secondary : (b), (c), (h); Tertiary : (f).
Question 4.
Explain why propanol has higher boiling point than that of the hydrocarbon, butane?
Answer:
Propanol undergoes intermolecular H-bonding because of the presence of -OH group. On the other hand, butane does not.
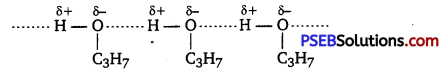
Therefore, extra energy is required to break hydrogen bonds. For this reason, propanol has a higher boiling point than hydrocarbon butane.
Question 5.
Alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses. Explain this fact.
Answer:
Alcohols form H-bonds with water due to the presence of -OH group. However, hydrocarbons cannot form H-bonds with water.
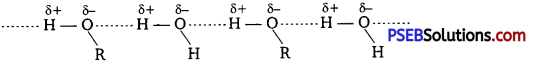
As a result, alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses.
Question 6.
What is meant by the hydroboration-oxidation reaction? Illustrate it with an example.
Answer:
The addition of diborane to alkenes to form trialkyl boranes followed by their oxidation with alkaline hydrogen peroxide to form alcohols is called hydroboration-oxidation reaction.
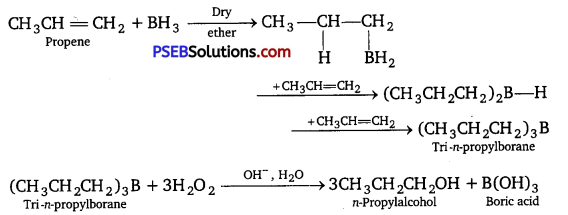
The alcohol obtained by this process is contrary to the Markovnikov’s rule.
Question 7.
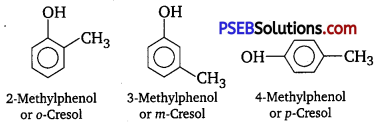
Give the structures and IUPAC names of monohydric phenols of molecular formula, C7H8O.
Answer:
The three isomers are given as follows :
Question 8.
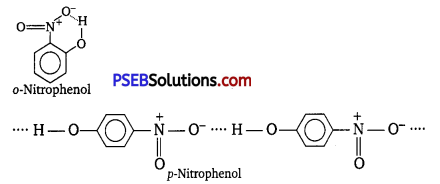
While separating a mixture of ortho and para-nitrophenols by steam distillation, name the isomer which will be steam volatile. Give reason.
Answer:
o-Nitrophenol is steam volatile as it exists as discrete molecules due to intramolecular H-bonding and hence can be separated by steam distillation from p-nitrophenol which is less volatile as it exists as associated molecules because of intermolecular H-bonding.
Question 9.
Give the equations of reactions for the preparation of phenol from cumene.
Answer:
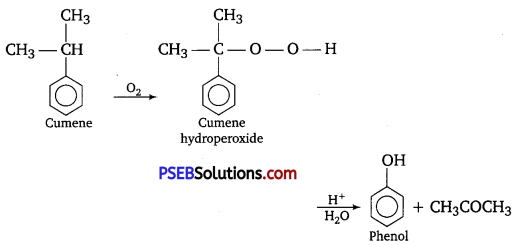
Question 10.
Write chemical reaction for the preparation of phenol from chlorobenzene.
Answer:
Chlorobenzene is fused with NaOH (at 623 K and 300 atm pressure) to produce sodium phenoxide, which gives phenol on acidification.
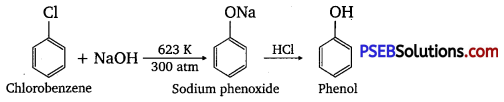
Question 11.
Write the mechanism of hydration of ethene to yield ethanol.
Answer:
The mechanism of hydration of ethene to yield ethanol involves three steps.
Step 1:
Protonation of ethene to form carbocation by electrophilic attack of H3O+:
H2O+H+ → H3O+
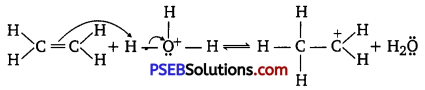
Step 2 :
Nucleophilic attack of water on carbocation:
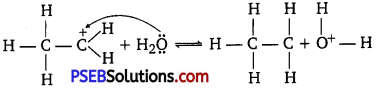
Step 3:
Deprotonation to form ethanol:
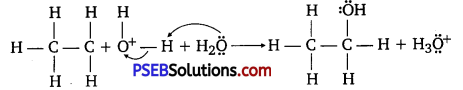
Question 12.
You are given benzene, cone. H2SO4 and NaOH. Write the equations for the preparation of phenol using these reagents.
Answer:
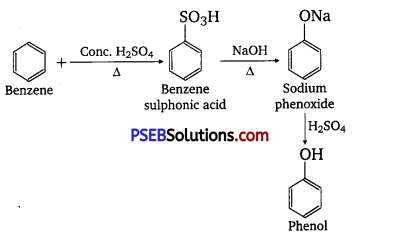
Question 13.
Show how will you synthesize:
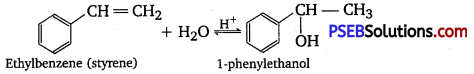
(i) 1-phenyl ethanol from a suitable alkene.
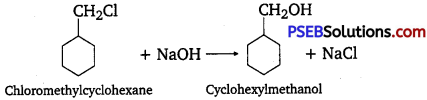
(ii) cyclohexyl methanol using an alkyl halide by an SN2 reaction.
(iii) pentane-l-ol using a suitable alkyl halide?
Answer:
(i) By acid-catalyzed hydration of ethylbenzene (styrene), 1-phenyl ethanol can be synthesized.
(ii) When chloro methylcyclohexane is treated with sodium hydroxide, cyclohexyl methanol is obtained.
(iii) When 1-chloromethane is treated with NaOH, pentane-l-ol is produced.
![]()
Question 14.
Give two reactions that show the acidic nature of phenol. Compare acidity of phenol with that of ethanol.
Answer:
The reactions showing acidic nature of phenol are :
(i) Reaction with sodium: Phenol reacts with active metals like sodium to liberate H2 gas.

(ii) Reaction with NaOH: Phenol dissolves in NaOH to form sodium phenoxide and water.
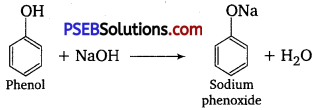
Comparison of acidic character of phenol and ethanol:
Phenol is more acidic than ethanol. This is due to the reason that phenoxide ion left after the loss of a proton from phenol is stabilized by resonance while ethoxide ion left after loss of a proton from ethanol is not stabilized by resonance.
Question 15.
Explain why is ortho nitrophenol more acidic than ortho methoxy phenol?
Answer:
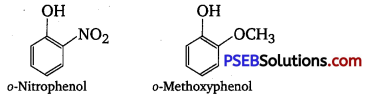
The nitro-group is an electron-withdrawing group. The presence of this group in the ortho position decreases the electron density in the O-H bond. As a result, it is easier to lose a proton. Also, the o-nitro phenoxide ion formed after the loss of protons is stabilized by resonance. Hence, ortho nitrophenol is a stronger acid. On the other hand, methoxy group is an electron-releasing group. Thus, it increases the electron density in the O-H bond and hence, the proton cannot be given out easily. For this reason,ortho-nitrophenol is more acidic than ortho- methoxy phenol.
Question 16.
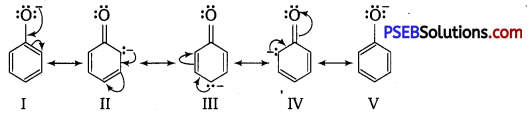
Explain how does the -OH group attached to a carbon of benzene ring activates it towards electrophilic substitution?
Answer:
Phenol may be regarded as a resonance hybrid of following structures:
Thus, due to +R effect of the -OH group, the electron density in the benzene ring increases thereby facilitating the attack of an electrophile. In other words, presence of -OH group, activates the benzene ring towards electrophilic substitution reactions. Now, since the electron density is relatively higher at the two o-and one p-position, electrophilic substitution occurs mainly at o-and p-positions.
Question 17.
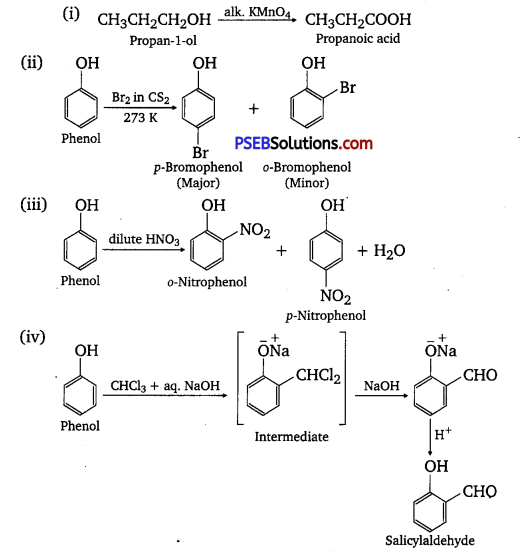
Give equations of the following reactions:
(i) Oxidation of propane-1-ol with alkaline KMnO4 solution.
(ii) Bromine in CS2 with phenol.
(iii) Dilute HNO3 with phenol.
(iv) Treating phenol with chloroform in presence of aqueous NaOH.
Answer:
Question 18.
Explain the following with an example.
(i) Kolbe’s reaction.
(ii) Reimer-Tlemimnn reaction.
(iii) WiIHamon ether synthesis.
(iv) Unsymmetrical ether.
Answer:
(i) Kolbe’s reaction: When phenol is treated with sodium hydroxide, sodium phenoxide is produced. This sodium phenoxide when treated with carbon dioxide, followed by acidification, undergoes electrophilic substitution to give ortho-hydroxybenzoic acid as the main product. This reaction is known as Kolbe’s reaction.
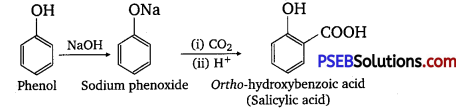
(ii) Reimer-Telemann reaction: When phenol is treated with chloroform (CHCl3 ) in the presence of sodium hydroxide, a -CHO group is introduced at the ortho position of the benzene ring.
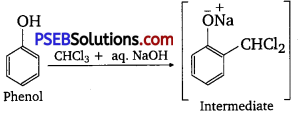
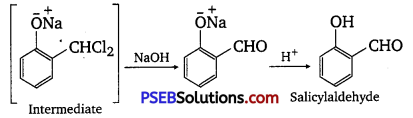
This reaction is known as the Reimer-Tiemann reaction. The intermediate is hydrolyzed in the presence of alkalis to produce salicylaldehyde.
(iii) Williamson ether synthesis: Williamson ether synthesis is a laboratory method to prepare symmetrical and unsymmetrical ethers by allowing alkyl halides to react with sodium alkoxides.
![]()
This reaction involves SN2 attack of the alkoxide ion on the alkyl halide. Better results are obtained in case of primary alkyl halides.
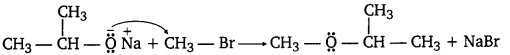
If the alkyl halide is secondary or tertiary, then elimination competes over substitution.
(iv) Unsymmetrical ether: An unsymmetrical ether is an ether where two groups on the two sides of an oxygen atom differ (i.e., have an unequal number of carbon atoms). For example: ethyl methyl ether (CH3-O-CH2CH3), methyl phenyl ether (CH3 -0 – C6H5), etc.
Question 19.
Write the mechanism of acid-catalyzed dehydration of ethanol to yield ethene.
Answer:
The mechanism of acid-catalyzed dehydration of ethanol to yield ethene involves the following three steps:
Step 1:
Protonaüon of ethanol to form ethyl oxonium ion:
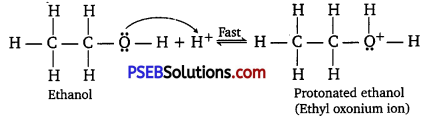
Step 2 :
Formation of carbocation (rate-determining step):

Step 3 :
Elimination of a proton to form ethene:
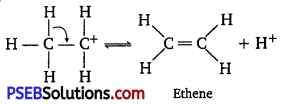
The acid consumed in Step 1 is released in Step 3. After the formation of ethene, it is removed to shift the equilibrium in a forward direction.
Question 20.
How are the following conversions carried out?
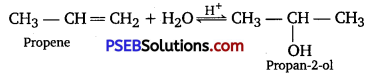
(i) Propene → Propan-2-ol
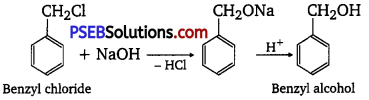
(ii) Benzyl chloride → Benzyl alcohol
(iii) Ethyl magnesium chloride → Propan-1-ol.
(iv) Methyl magnesium bromide → 2-Methylpropan-2-ol.
Answer:
(i) If propene is allowed to react with water in the presence of an acid as a catalyst, then propane-2-ol is obtained.
(ii) If benzyl chloride is treated with NaOH (followed by acidification) then benzyl alcohol is produced.
(iii) When ethyl magnesium chloride is treated with methanal, an adduct is produced which gives propane-1-ol on hydrolysis.
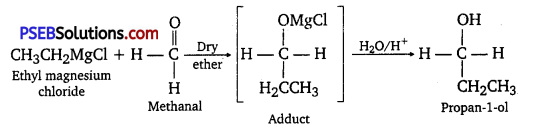
(iv) When methyl magnesium bromide is treated with propanone, an adduct is produced which gives 2- methylpropane-2-ol on hydrolysis.
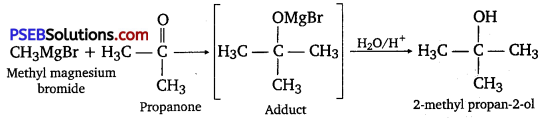
Question 21.
Name the reagents used in the following reactions:
(i) Oxidation of a primary alcohol to carboxylic acid.
(ii) Oxidation of a primary alcohol to aldehyde.
(iii) Bromination of phenol to 2,4,6-tribromophenol.
(iv) Benzyl alcohol to benzoic acid.
(v) Dehydration of propane-2-ol to propene.
(vi) Butan-2-one to butan-2ol.
Answer:
(i) Acidified potassium dichromate (K2Cr2O7/H2SO4)
(ii) Pyridinium chlorochromate (PCC)
(iii) Bromine water (Br2/H2O)
(iv) Acidified potassium permanganate (KMnO4/H2SO4)
(v) 85% phosphoric acid (H3PO4)
(vi) NaBH4 or LiAlH4.
Question 22.
Give reason for the higher boiling point of ethanol in comparison to methoxymethane.
Answer:
Ethanol undergoes intermolecular H-bonding due to the presence of -OH group, resulting in the association of molecules. Extra energy is required to break these hydrogen bonds. On the other hand, methoxymethane does not undergo H-bonding. Hence, the boiling point of ethanol is higher than that of methoxymethane.

Question 23.
Give IUPAC mimes of the following ethers:
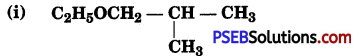
(ii) CH3OCH2CH2Cl
(iii) O2N- C6H4 – OCH3(p)
(iv) CH3CH2CH2OCH3
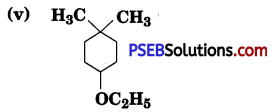
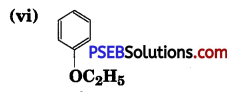
Answer:
(i) 1-Ethoxy-2-methylpropane
(ii) 2-Chioro- 1 -methoxyethane
(iii) 4-Nitroanisole
(iv) 1 -Methoxypropane
(v) 4-Ethoxy- 1, 1 -dime thylcyclohexane
(vi) Ethoxybenzene.
Question 24.
Write the names of reagents and equations for the preparation of the following ethers by WilÌianlÑon’s synthesis:
(i) 1-Propoxypropane
(ii) Ethoxybenzene
(iii) 2.Methoxy-2-methylpropane
(iv) 1 -Methoxyethane
Answer:
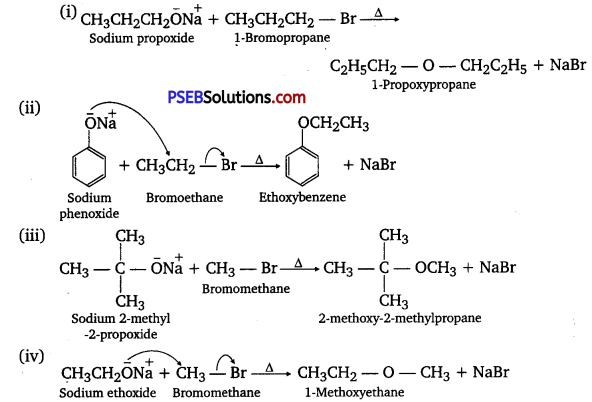
Question 25.
Illustrate with examples the limitations of Willi9mon synthesis for the preparation of certain types of ethers.
Answer:
The reaction of Williamson synthesis involves SN2 attack of an alkoxide ion on a primary alkyl halide.

But if secondary or tertiary alkyl halides are taken in place of primary alkyl halides, then elimination would compete over substitution. As a result, alkenes would be produced. This is because alkoxides are nucleophiles as well as strong bases. Hence, they react with alkyl halides, which results in an elimination reaction.
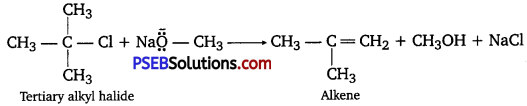
Question 26.
How is 1-propoxy propane synthesized from propane-l-ol? Write mechanism of this reaction.
Answer:
1-propoxy propane can be synthesized from propane-1-ol by dehydration Propan- 1 -ol undergoes dehydration in the presence of protic acids (such as H2SO4, H3PO4) to give 1-propoxy propane.
![]()
The mechanism of this reaction involves the following three steps:
Step 1: Protonation

Step 2: Nucleophilic attack
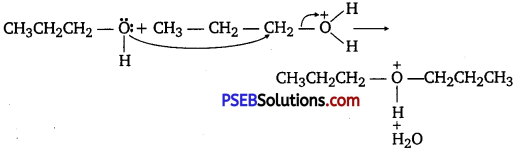
Step 3: Deprotonanon
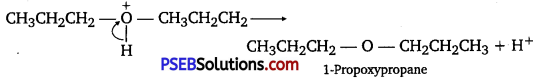
Question 27.
Preparation of ethers by acid-catalyzed dehydration of secondary or tertiary alcohols is not a suitable method. Give
reason.
Answer:
Acid-catalyzed dehydration of primary alcohols to ethers occurs by SN2 reaction involving nucleophilic attack of the alcohol molecule on the protonated alcohol molecule.
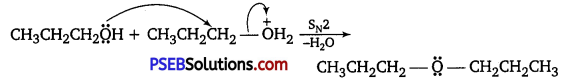
Under these conditions, secondary and tertiary alcohols, however, give alkenes rather than ethers. This is because due to steric hindrance, nucleophilic attack of the alcohol molecule on the protonated alcohol molecule does not occur. Instead, protonated secondary and tertiary alcohols lose a molecule of water to form stable secondary and tertiary carbocations. These carbocations prefer to lose a proton to form alkenes rather than undergoing nucleophilic attack by alcohol molecules to form ethers.
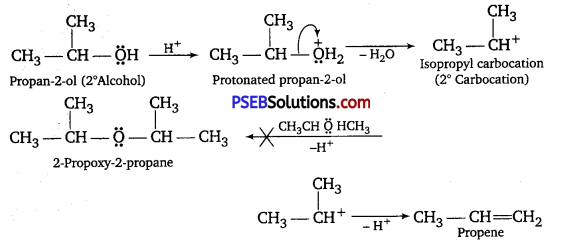
Similarly, tertiary alcohols give alkenes rather than ethers.
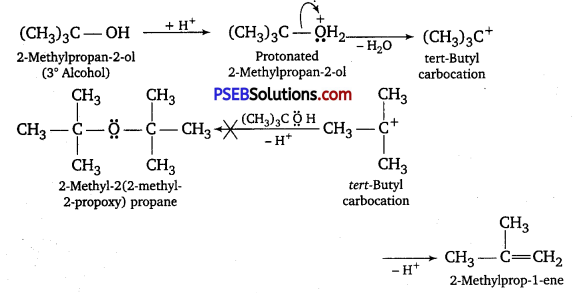
Question 28.
Write the equation of the reaction of hydrogen iodide with:
(i) 1-propoxy propane
(ii) Methoxybenzene and
(iii) Benzyl ethyl ether
Answer:
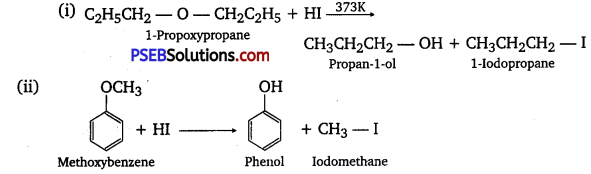
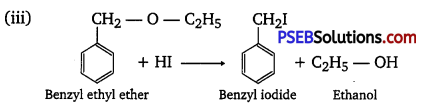
Question 29.
ExplaIn the fact that in aryl alkyl ethers
(i) the alkoxy group activates the benzene ring towards electrophilic substitution and
(ii) it directs the incoming substituents to ortho and para positions in benzene ring.
Answer:
(i) In aryl alkyl ethers, the +Reffect of the alkoxy group (OR) increases the electron density in the benzene ring thereby activating the benzene ring towards electrophilic substitution reactions.
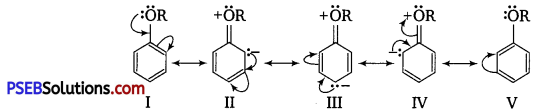
(ii) Since the electron density increases more at the two orthos and one para position as compared to meta-positions, electrophilic substitution reactions mainly occur at ortho-and para-positions. For example,
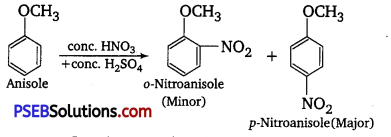
Question 30.
Write the mechanism of the reaction of III with methoxymethane.
Answer:
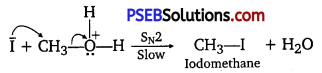
When equimolar amounts of HI and methoxymethane are taken; a mixture of methyl alcohol and iodomethane are formed.
Mechanism
Step I:

Step II :
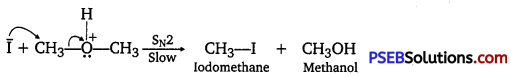
If HI is present in excess, CH3OH formed in step II is further converted into CH3I.
Step III:

Step IV:
Question 31.
Write equations of the following reactions:
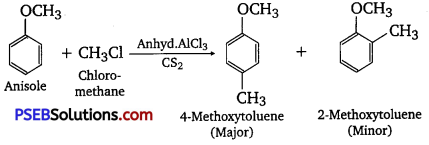
(i) Friedel-Crafts reaction-alkylation of anisole.
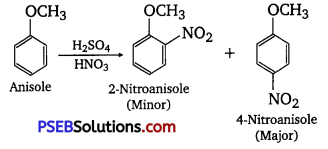
(ii) Nitration of anisole.
(iii) Bromlnation of anisole in ethanoic acid medium.
(iv) Friedel-Craft’s acetylation of anisole.
Answer:
(i) Friedel-Crafts reaction (Alkylation of anisole)
(ii) Nitration of anisole
(iii) Bromination of anisole in ethanoic acid medium
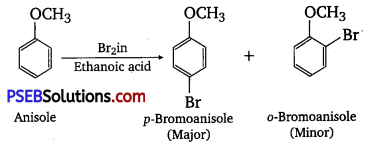
(iv) Friedel-Crafts acetylation of anisole
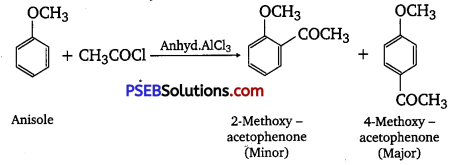
Question 32.
Show ho would you synthesize the following alcohols from appropriate alkenes?

Answer:
The given alcohols can be synthesized by applying Markovnikov’s rule of acid-catalyzed hydration of appropriate alkenes. –
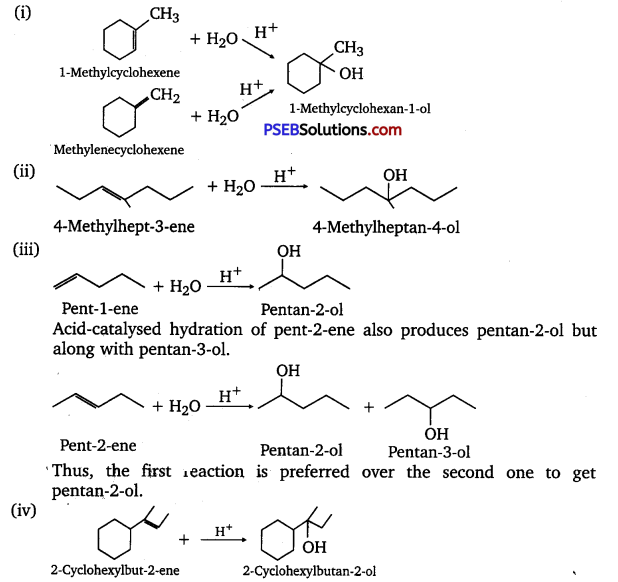
Question 33.
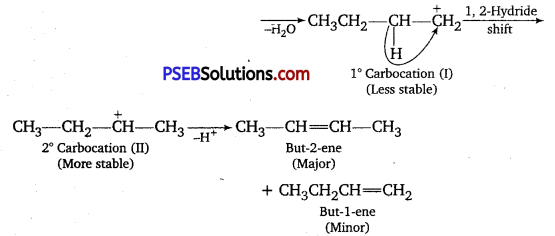
When 3-methyl butan-2-ol is treated with HBr, the following reaction takes place:
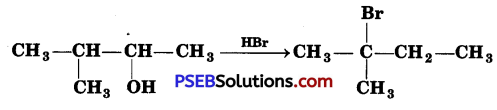
Give a mechanism for this reaction.
(Hint: The secondary carbocation formed in step It rearranges to a more stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.)
Answer:
Mechanism: The reaction takes place through the following mechanism :
Step I: Formation of protonated alcohol.
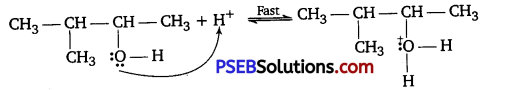
Step II: Formation of carbocatlon.
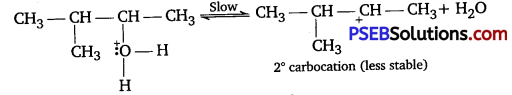
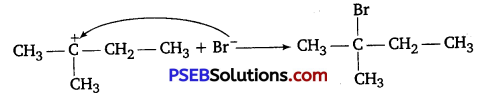
2° carbocation being less stable undergoes hydride shift to form more stable 3° carbocation.

Step III: Attack of nucleophile
Chemistry Guide for Class 12 PSEB Alcohols, Phenols, and Ethers Textbook Questions and Answers
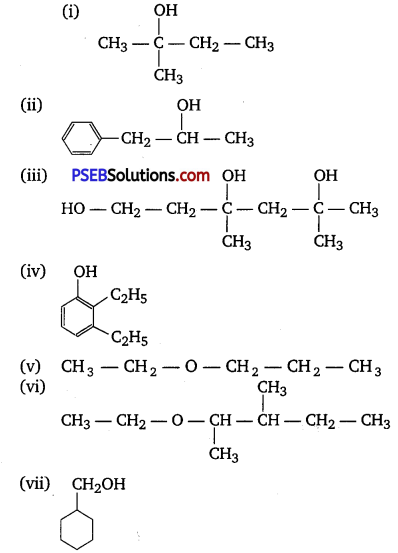
Question 1.
Classify the following as primary, secondary and tertiary alcohols:
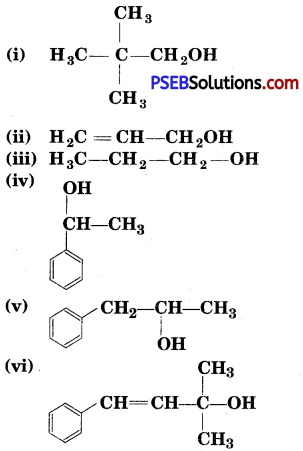
Answer:
Primary alcohols (i), (ii), (iii)
Secondary alcohols (iv) and (v)
Tertiary alcohols (vi)
Question 2.
Identify allylic alcohols in the above examples.
Answer:
Allylic alcohols (ii) and (vi).
Question 3.
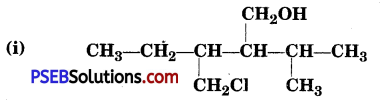
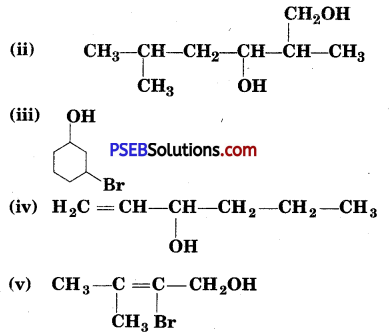
Name the following compounds according to IUPAC system :
Answer:
(i) 3-Chloromethyl-2-isopropylpentan-l-ol _
(ii) 2, 5-Dimethylhexane-l,3-diol .
(iii) 3-Bromocyclohexanol
(iv) Hex-l-en-3-ol
(v) 2-Bromo-3-methylbut-2-en-l-ol.
Question 4.
Show how are the following alcohols prepared by the reaction of a suitable Grignard reagent on methanal.
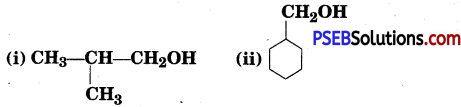
Answer:
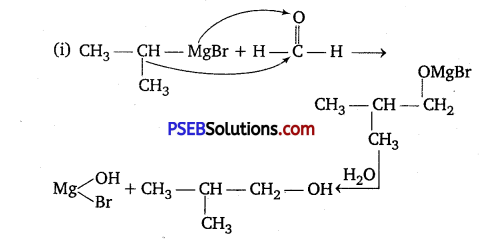
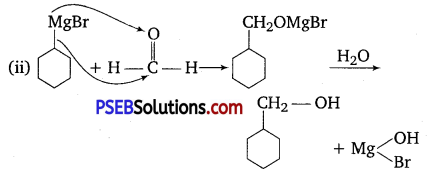
Question 5.
Write structures of the products of the following reactions:
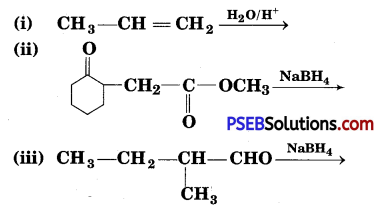
Answer:
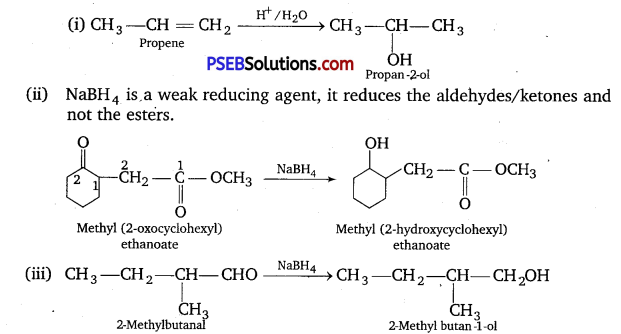
Question 6.
Give structures of the products you would expect when each of the following alcohol reacts with
(a) HCl-ZnCl2
(b) HBr and
(c) SOCl2.
Answer:
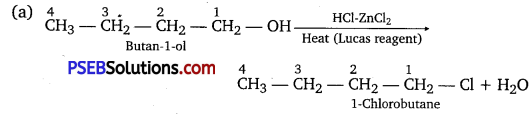
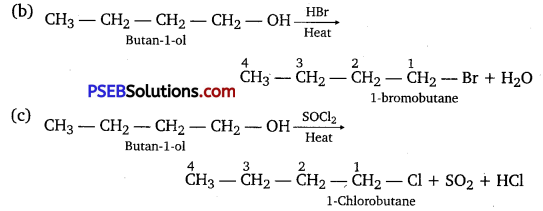
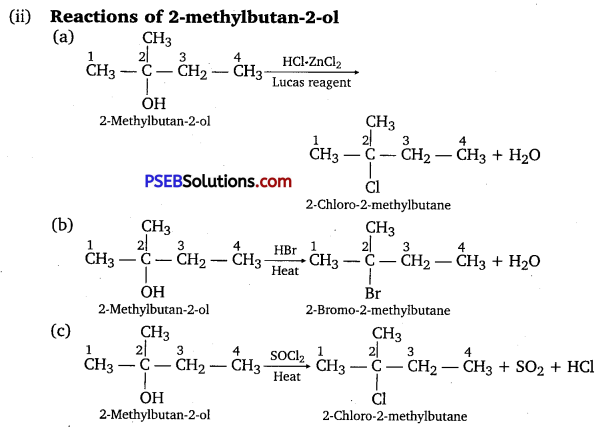
Question 7.
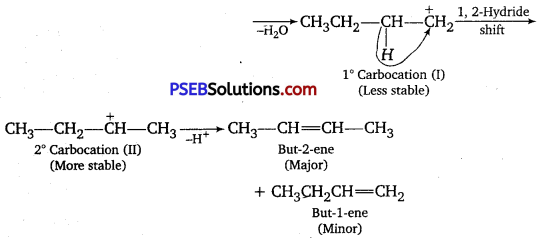
Predict the major product of acid-catalyzed dehydration of
(i) 1-methyl cyclohexanol and
(ii) butane-l-ol.
Answer:
(i) Acid-catalyzed dehydration of 1-methyl cyclohexanol can give two products, I and II. Since product (I) is more substituted, according to Saytzeff rule, it is the major product.
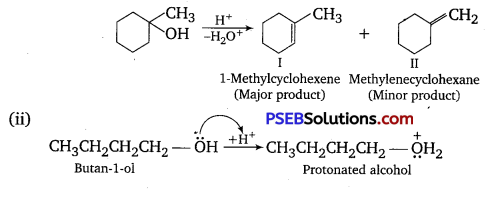
Question 8.
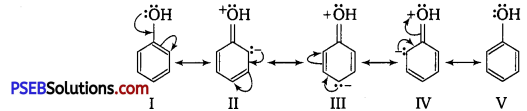
Ortho and para-nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Answer:
The resonance structures of o – and p -nitrophenoxide ions and phenoxide ion are given as follows :
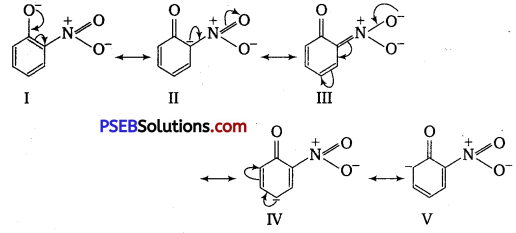
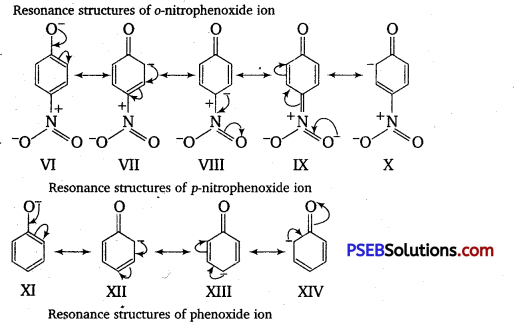
It is clear from the above structures that due to -R effect of NO2 group, o-and p-nitro phenoxide ions are more stable than phenoxide ions. Consequently, o-and p-nitrophenols are more acidic than phenol.
Question 9.
Write the equations involved in the following reactions :
(i) Reimer-Tiemann reaction
(ii) Kolbe’s reaction –
Answer:
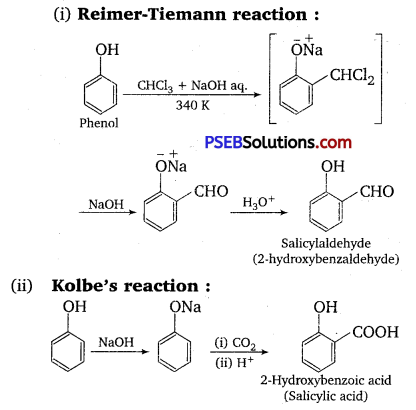
Question 10.
Write the reactions of Williamson synthesis of 2- ethoxy-3-methyl pentane starting from ethanol and 3- methyl pentane-2-ol.
Answer:
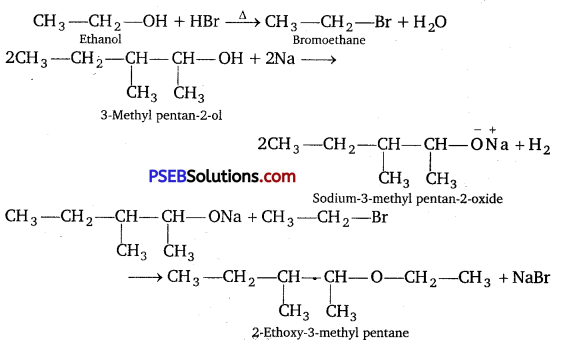
Question 11.
Which of the following is an appropriate set of reactants for the preparation of l-methoxy-4- nitrobenzene and why ?
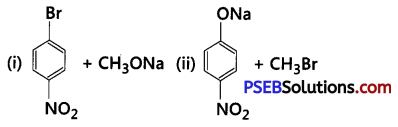
Answer:
(ii) because inset (i), sodium methoxide (CH3ONa) is a strong nucleophile as well as a strong base. So, elimination reaction predominates over substitution reaction.
Question 12.
Predict the products of the following reactions :
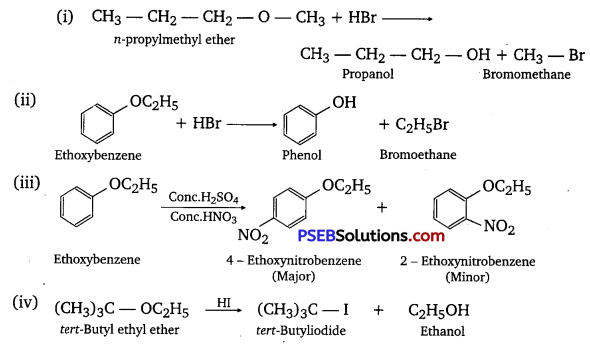
(i) CH3 – CH2 – CH2 – O – CH3 + HBr →
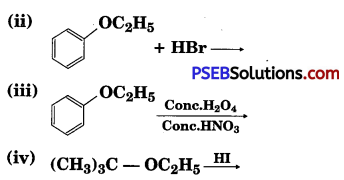
Answer: