Punjab State Board PSEB 11th Class Biology Book Solutions Chapter 9 Biomolecules Textbook Exercise Questions and Answers.
PSEB Solutions for Class 11 Biology Chapter 9 Biomolecules
PSEB 11th Class Biology Guide Biomolecules Textbook Questions and Answers
Question 1.
What are macromolecules? Give examples.
Answer:
Chemical compounds, which are found in the acid insoluble fraction are called macromolecules or biomacromolecules. For example, proteins, lipids and carbohydrate, etc.
Question 2.
Illustrate a glycosidic, peptide and a phosphodiester bond.
Answer:
Glycosidic Bond: A glycosidic bond is a type of functional group that joins a carbohydrate (sugar) molecule to another group, which may or may not be another carbohydrate.
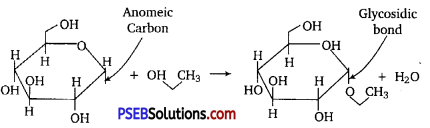
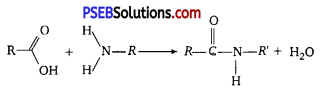
Peptide Bond: A peptide bond (amide bond) is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amine group of the other molecule, thereby releasing a molecule of water (H20).
H2O.
Phosphodiester Bond: A phosphodiester bond is a group of strong covalent bonds between a phosphate group and two other molecules over two ester bonds. In DNA and RNA, the phosphodiester bond is the linkage between the 3′ carbon atom of one sugar molecule and the 5’carbon of another, deoxyribose in DNA and ribose in RNA.
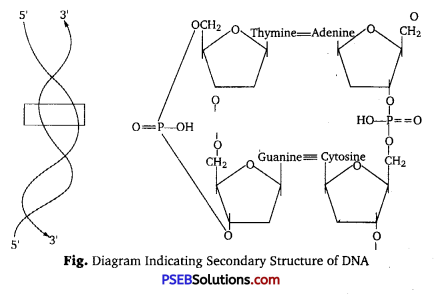
![]()
Question 3.
What is meant by tertiary structure of proteins?
Answer:
Tertiary Structure of Protein; The overall shape of a single protein molecule; the spatial relationship of the secondary structures to one another. Tertiary structure is generally stabilized by non-local interactions, most commonly the formation of a hydrophobic core, but also through salt bridges, hydrogen bonds, disulfide bonds,1 and even post-translational modifications. The term “tertiary structure” is often used as synonymous with the term fold. The tertiary structure is what controls the basic function of the protein.
Question 4.
Find and write down structures of 10 interesting small molecular weight biomolecules. Find if there is any industry which manufactures the compounds by isolation. Find out who are the buyers?
Answer:
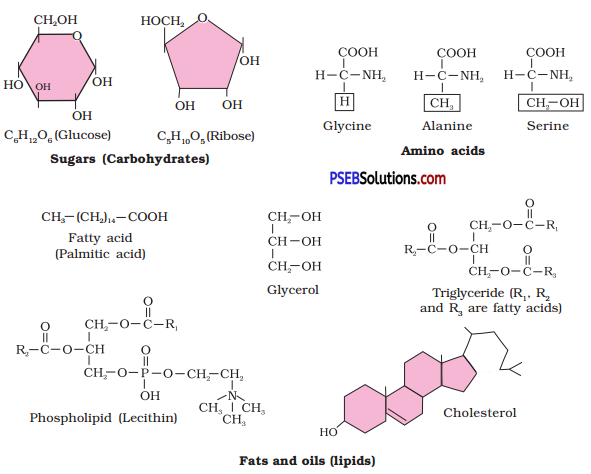
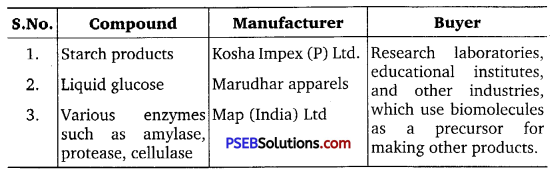
Question 5.
Proteins have primary structure. If you are given a method to know which amino acid is at either of the two termini (ends) of a protein, can you connect this information to purity or homogeneity of a protein?
Answer:
The sequence of amino acids, i.e., the positional information in a protein which is the first amino acid, which is second and so on is called the primary structure of a protein. The first amino acid is also called as N-terminal amino acid. The last amino acid is called the C-terminal amino acid. Yes, we can connect this information to purity or homogeneity of a protein. Based on number of amino and carboxyl groups, there are acidic (e.g., glutamic acid), basic (lysine) and neutral (valine) amino acids, proteins may be acidic, basic and neutral.
![]()
Question 6.
Find out and make a list of proteins used as therapeutic agents. Find other applications of proteins (e. g, cosmetics etc.)
Answer:
Some proteins and their functions are as follows:
| Proteins | Functions |
| 1. Collagen | Intercellular ground substance |
| 2. Trypsin | Enzyme |
| 3. Insulin | Hormone |
| 4. Antibody | Fights against infections |
| 5. Receptors | Sensory reception (example-taste) |
| 6. Glut-4 | Enables glucose transport in cells |
| 7. Keratolytic protein | Used to soften hard skin |
| 8. Egg protein | Used for skin tightening |
Question 7.
Explain the composition of triglyceride.
Answer:
Triglycerides are composed of two types of molecules, i.e., glycerol i (3 carbon molecules) and fatty acids which attach to the glycerol at the alcohol unit. The following is a structural representation of a triglyceride at the molecular level.
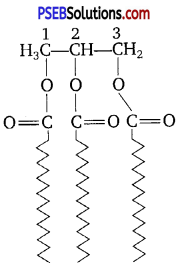
Fatty acids are chains of hydrocarbons 4-22 (or more) carbons a long with a carboxyl group at one end. If each carbon has two hydrogen atoms, the fatty acid is saturated. If two carbon atoms are double-bonded, so that there is less hydrogen in the fatty acid, it is unsaturated (monounsaturated). If more than two carbon atoms are unsaturated, the fatty acid is polyunsaturated.
Question 8.
Can you describe what happens when milk is converted into curd or yoghurt, from your understanding of proteins.
Answer:
Milk contains a protein called casein. This protein gives milk its characteristic white colour. It is of high nutritional value because it contains all the essential amino acids required by man’s body. The curd forms because of the chemical reaction between lactic acid bacteria and casein. When curd is added to milk, the lactic acid bacteria present in it cause coagulation of casein and thus, convert it into curd.
Question 9.
Can you attempt building models of biomolecules using commercially available atomic models (Ball and Stick models).
Answer:
Yes, we can make models of biomolecules using commercially available atomic models.
![]()
Question 10.
Attempt titrating an amino acid against a weak base and discover the number of dissociating (ionisable) functional groups in the amino acid.
Answer:
When an amino acid is titrated against a weak base, it dissociates and gives two functional groups:
(i) -COOH group (carboxylic group)
(ii) Amino group (NH2/sub>)
Question 11.
Draw the structure of the amino acid, alanine.
Answer:
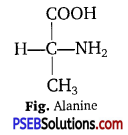
Question 12.
What are gums made of? Is fevicol different?
Answer:
Gums are made of carbohydrates, i. e., L-rhamnose, D-galactose and D-galacturonic acid, etc. Fevicol is different from natural gums. It is a synthetic product.
Question 13.
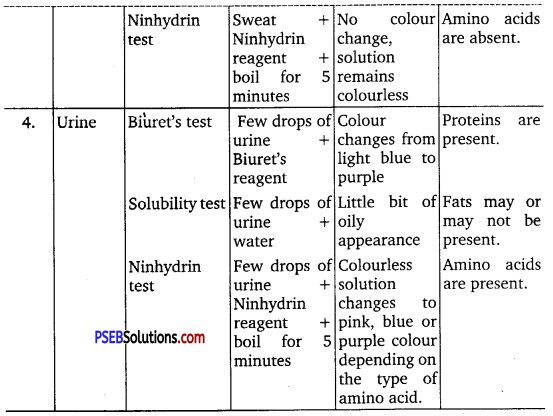
Find out a qualitative test for proteins, fats and oils, amino acids and test any Fruit juice, saliva, sweat and urine for them.
Answer:
- Test for Proteins: Biuret test if Biuret’s reagent added to protein, then the colour of the reagent changes light blue to purple.
- Test for Fats and Oils: Grease or test.
- Test for Amino Acids: Ninhydrin test. If Ninhydrin reagent is added to the solution, then the colourless solution changes to pink, blue or purple depending upon the amino acid.
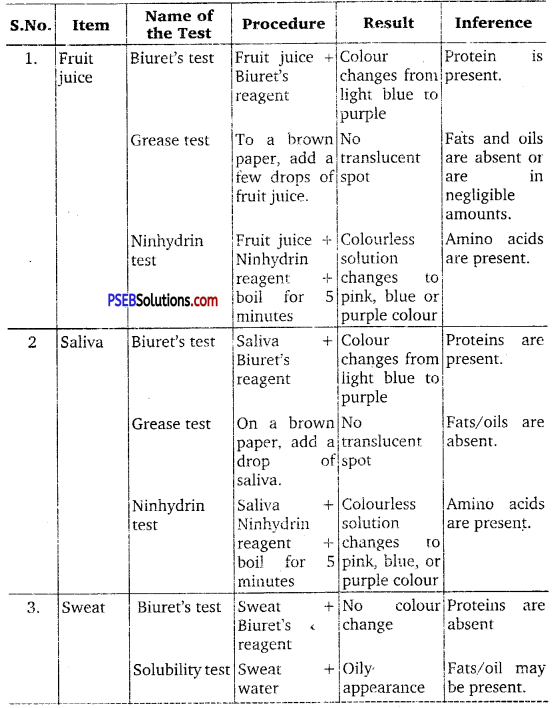
![]()
Question 14.
Find out how much cellulose is made by all the plants in the biosphere and compare it with how much of paper is manufactured by man and hence what is the consumption of plant material by man annually. What a loss of vegetation!
Answer:
Approximately, 100 billion tonnes of cellulose are made per year by all the plants in the biosphere and it takes 17 full grown trees to make one ton of paper. Trees are also used to fulfil the other requirements of man such as for timber, food, medicines, etc. Hence, it is difficult to calculate the annual consumption of plant material by man.
Question 15.
Describe the important properties of enzymes.
Answer:
Important properties of enzymes are given below :
- Enzymes are proteins which catalyse biochemical reactions in the cells.
- They are denatured at high temperatures.
- Enzymes generally function in a narrow range of temperature and pH. Each enzyme shows its highest activity at a particular temperature and pH called the optimum temperature and optimum pH.
- With the increase in substrate concentration, the velocity of the eyzymatic reaction rises at first. The reaction ultimately reaches a maximum velocity (Vmax) which is not exceeded by any further rise in concentration of the
substrate. - The activity of an enzyme is also sensitive to the presence of specific chemicals that bind to the enzyme.
- Enzymes are substrate specific in their action.