Punjab State Board PSEB 11th Class Chemistry Important Questions Chapter 12 Organic Chemistry: Some Basic Principles and Techniques Important Questions and Answers.
PSEB 11th Class Chemistry Important Questions Chapter 12 Organic Chemistry: Some Basic Principles and Techniques
Very Short Answer Type Questions
Question 1.
How many σ bonds and π bonds are present in the second member of the alkene series?
Answer:
The second member of the alkene series is propene. The structual formula of the propene is
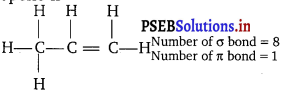
Question 2.
Show the polarisation of carbon-magnesium bond in the following structure.
CH3 —CH2 —CH2 —CH2 —Mg —X
Answer:
Carbon is more electronegative than magnesium. Therefore, Mg acquires a partial positive charge and carbon acquires a partial negative charge.
![]()
![]()
Question 3.
What are primary and secondary suffixes as applied to IUPAC nomenclature?
Answer:
The primary suffix indicates whether the carbon chain is saturated or unsaturated while the secondary suffix indicates the functional group present in the molecule.
Question 4.
Draw all position isomers of an alcohol with molecular formula, C3HgO.
Answer:
![]()
Question 5.
CH2 = CH– is more basic than HC = C– . Explain why?
Answer:
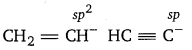
Since, sp-carbon is more electronegative than sp2-carbon, therefore, CH ≡ C– is less willing to donate a pair of electrons than H2C = CH–. In other words, H2C = CH– is more basic than HC ≡ C–.
Question 6.
Why does SO3 act as an electrophile? [NCERT Exemplar]
Answer:
In SO3, three highly electronegative oxygen atoms are attached to sulphur atom. It makes sulphur atom electron deficient. Further, due to resonance, sulphur acquires a positive charge. Both these factors, make SO3 an electrophile.
Question 7.
How will you separate a mixture of o-nitro phenol and p-nitrophenol?
Answer:
A mixture of o-nitrophenol and p-nitrophenol can be separated by steam distillation, o-nitrophenol being less volatile distils over along with water while p-nitrophenol being non-volatile remains in the flask.
Question 8.
In DNA and RNA, nitrogen atom is present in the ring system. Can Kjeldahl’s method be used for the estimation of nitrogen present in these? Give reason.
Answer:
DNA and RNA have nitrogen in the heterocyclic rings. Nitrogen present in rings, azo and nitro groups cannot be converted into (NH4)2SO4. That’s why Kjeldahl’s method cannot be used for the estimation of nitrogen present in these.
Question 9.
Lassaigne’s test is not shown by diazonium salts, though they contain nitrogen. Why?
Answer:
Diazonium salts (C6H5N2+X–) readily lose N2 on heating before reacting with fused sodium metal. Therefore, these salts do not give positive Lassaigne’s test for nitrogen.
![]()
Question 10.
Write three-dimensional wedge-dashed or wedge-line representations for the following:
(a) CH3CH2OH
(b) CH2FCl
Answer:
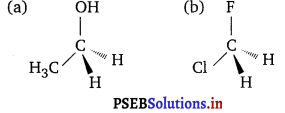
Short Answer Type Questions
Question 1.
Draw all polygon formula for the molecular formula C5H10.
Answer:
The different polygon formula of the compound having molecular formula C5H10 are :
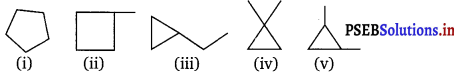
Question 2.
An alkane has a molecular mass of 72. Draw all its possible chain isomers and write their IUPAC names.
Answer:
First of all, we will derive the molecular formula.
Molecular formula of alkane is CnH2n+2
∴ Molecular mass = 72
∴ 12n + 2n + 2 = 72
n = 5
The alkane is C5H12. The possible chain isomers are
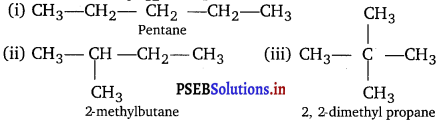
Question 3.
Arrange the following

Answer:
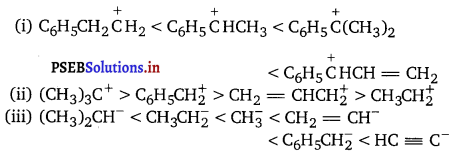
![]()
Question 4.
Suggest a method to purify
(i) camphor containing traces of common salt.
(ii) kerosene oil containing water.
(iii) a liquid which decomposes at its boiling point.
Answer:
(i) Sublimation-camphor sublimes while common salt remains as residue in the China dish.
(ii) Since the two liquids are immiscible, the technique of solvent extraction with a separating funnel is used. The mixture is thoroughly shaken and the separating funnel is allowed to stand. Kerosene being lighter than water forms the upper layer while water forms the lower layer.
The lower water layer is run off using the stop cork of the funnel and kerosene oil is obtained. It is dried over anhydrous CaCl2 or MgSO4 and then distilled to give pure kerosene oil.
(iii) Distillation under reduced pressure.
Question 5.
The structure of triphenylmethyl cation is given below. This is very stable and some of its salts can he stored for months. Explain the cause of high stability of this cation.
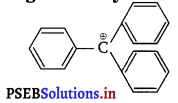
Answer:
In triphenylmethyl cation, due to resonance, the positive charge can move at both the o-and p-position of each benzene ring. This is illustrated below.
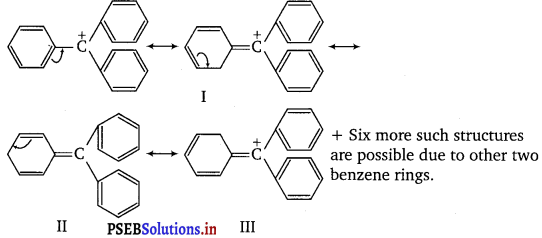
Since, there are three benzene rings, hence, there are, in all, nine resonance structures.
Thus, triphenylmethyl cation is highly stable due to these nine resonance structures.
Long Answer Type Questions
Question 1.
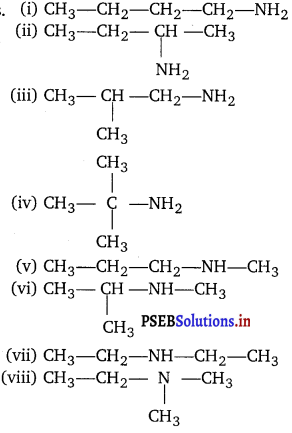
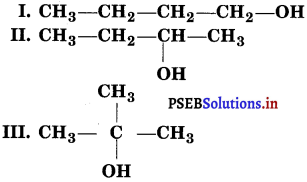
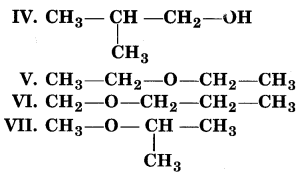
Consider structures I to VII and answer the following question (i) to (iv).
(i) Which of the above compounds form pairs of metamers?
(ii) Identify the pairs of compounds which are functional group isomers.
(iii) Identify the pairs of compounds that represent position isomerism.
(iv) Identify the pairs of compounds that represent chain isomerism.
Answer:
(i) V and VI or VI and VII form a pair of metamers since they differ in the number of carbon atoms on the either side of the functional group, i.e., O-atom.
(ii) I and V, I and VI, I and VII; II and V, II and VI, II and VII; III and V, III and VI; III and VII; IV and V; IV and VI and IV and VII are all functional group isomers.
(iii) I and II, III and IV and, VI and VII represent position isomerism.
(iv) I and III, I and IV, II and III and II and IV represent chain isomerism.
![]()
Question 2.
Write structural formulae for all the isomeric amines with molecular formula C^^N.
Answer: