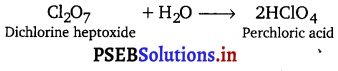Punjab State Board PSEB 11th Class Chemistry Important Questions Chapter 3 Classification of Elements and Periodicity in Properties Important Questions and Answers.
PSEB 11th Class Chemistry Important Questions Chapter 3 Classification of Elements and Periodicity in Properties
Very Short Answer Type Questions
Question 1.
Which properties of the elements depend on’ the electronic configuration of the atoms and which do not?
Answer:
Chemical and many physical properties of the elements depends on the electronic configuration of the atoms, whereas the nuclear properties do not.
Question 2.
Write the number designation of a group that has 2 electrons beyond a noble gas configuration.
Answer:
The number designation of a group that has 2 electrons beyond a noble gas configuration will be 2 which means it will belong to group 2 of periodic table.
Question 3.
Why is it more logical to call the atomic radius as the effective atomic radius?
Answer:
This is because the size of atom is very small and it has no sharp boundaries.
![]()
Question 4.
A boy has reported the radii of Cu, Cu+ and Cu2+ as 0.096 nm, 0.122 nm and Question072 nm respectively. However, it has been noticed that he interchanged the values by mistake. Assign the correct values to different species.
Answer:
Cu [0.122 nm], Cu+ [0.096 nm], Cu2+ [0.072 nm].
∵ Size ∝ \(\frac{1}{\text { positive charge }}\)
Question 5.
Atomic radii of fluorine is 72 pm where as atomic radii of neon is 160 pm. Why? [NCERT Exemplar]
Answer:
Atomic radius of F is expressed in terms of covalent radius while, atomic radius of neon is usually expressed in terms of van der Waals’ radius, van der Waals’ radius of an element is always larger than its covalent radius.
Therefore, atomic radius of F is smaller than atomic radius of Ne (F = 72 pm, Ne = 160 pm)
Question 6.
Arrange the following elements in order of decreasing electron gain enthalpy : B, C, N, O.
Answer:
N has positive electron gain enthalpy while all others have negative electron gain enthalpies. Since size decreases on moving from B → C → O, therefore, electron gain enthalpies become more and more negative from B → C → O. Thus, the overall decreasing order of electron gain enthalpies is N, B, C, O.
Question 7.
Which of the following atoms would most likely form an anion (i) Be, (ii) Al, (iii) Ga, (iv) I ?
Answer:
I, because of high electron gain enthalpy, it can accept an electron readily to form an anion F < Cl < Br > I.
Question 8.
Explain why chlorine can be converted into chloride ion more easily as compared to fluoride ion from fluorine.
Answer:
Electron gain enthalpy of Cl is more negative than that of F.
Question 9.
Among alkali metals which element do you expect to be least electronegative and why? [NCERT Exemplar]
Answer:
On moving down the group, electronegativity decreases because atomic size increases. Fr has the largest size, therefore it is least electronegative.
Question 10.
Arrange the following elements in the increasing order of non-metallic character. B, C, Si, N, F
Answer:
The given non-metals are arranged in the increasing order of non-metallic character as follows:
![]()
![]()
Short Answer Type Questions
Question 1.
What would be IUPAC names and symbols for elements with atomic numbers 122, 127, 135, 149 and 150? .
Answer:
The roots 2, 7, 5, 9 and 0 are referred as bi, sept, pent, enn and nil respectively. Therefore, their names and symbol are
| Z (Atomic number) | Name | Symbol |
| 122 | Unbibium | Ubb |
| 127 | Unbiseptium | Ubs |
| 135 | Untripentium | Dtp |
| 149 | Unquadennium | Uqe |
| 150 | Unpentnilium | Upn |
Question2.
All transition elements are d-block elements, but all d-block elements are not transition elements. Explain.
Answer:
Elements in which the last electron enters in the d-orbitals, are called d-block elements or transition elements. These elements have the general outer electronic configuration (n – 1)d1-10ns0-2 Zn, Cd and Hg having the electronic configuration, (n – l1)d10ns2 do not show most of the properties of transition elements. The d-orbitals in these elements are completely filled in the ground state as well as in their common oxidation states. Therefore, they are not regarded as transition elements. Thus, on the basis of properties, all transition elements are d-block elements but on the basis of electronic configuration, all d-block elements are not transition elements.
Question 3.
Arrange the elements N, P, O and S in the order of
(i) increasing first ionisation enthalpy.
(ii) increasing non-metallic character.
Give reason for the arrangement assigned.
Answer:
| Group 15 | Group 16 | |
| 2nd period | N | 0 |
| 3rd period | P | S |
(i) Ionisation enthalpy of nitrogen (7N = 1s2, 2s2, 2p3) is greater than oxygen (8O = 1s2 , 2s2 , 2p4 ) due to extra stable half-filled 2p-orbitals. Similarly, ionisation enthalpy of phosphorus (15P = 1s2, 2s2, 2p6, 3s2, 3p3) is greater than sulphur (16S = 1s2, 2x2, 2p6, 3s2, 3p4).
On moving down the group, ionisation enthalpy decreases with increasing atomic size. So, the increasing order of first ionisation enthalpy is S < P < O < N.
(ii) Non-metallic character across a period (left to right) increases but on moving down the group it decreases. So, the increasing order of non-metallic character is P < S < N < 0.
![]()
Question 4.
What do you understand by exothermic reaction and endothermic reaction? Give one example of each type.
Answer:
Exothermic reactions : Reactions which are accompanied by evolution of heat are called exothermic reactions. The quantity of heat produced is shown either along with the products with a ‘+’ sign or in terms if ΔH with a sign, e.g.,
C(s) + O2(g) → CO2(g) + 393.5 kJ
H2(g) + \(\frac{1}{2}\)O2(g) → H2O(l) ΔH = -285.8 kJ mol-1
Endothermic reactions : Reactions which proceed with absorption of heat are called endothermic reactions. The quantity of heat absorbed is shown either alongwith the products with a sign or in terms of ΔH with a ‘-‘ sign, e.g.,
C(s) + H2O(g) → CO(g) + H2(g) -131.4 kJ
N2(g) + 3H2(g) → 2NH3(g); ΔH = +92.4 kJ mol-1
Question5.
How does the metallic and non-metallic character vary on moving from left to right in a period?
Answer:
As we move from left to right in a period, the number of valence electrons increases by one at each succeeding element but the number of shells remains same. Due to this, effective nuclear charge increases.
More is the effective nuclear charge, more is the attraction between nuclei and electron.
Hence, the tendency of the element to lose electrons decreases, this results in decrease in metallic character.
Furthermore, the tendency of an element to gain electrons increases with increase in effective nuclear charge, so non-metallic character increases on moving from left to right in a period.
Long Answer Type Questions
Question 1.
Write the drawbacks in Mendeleev’s Periodic Table that led to its modification.
Answer:
The main drawbacks of Mendeleev’s Periodic Table are:
(i) Some elements having similar properties were placed in different groups whereas some elements having dissimilar properties were placed in the same group. For example alkali metals such as Li, Na, K, etc., (I A group) are grouped together with coinage metals such as Cu, Ag, Au (I B group) though their properties are quite different. Chemically similar elements such as Cu (I B group) and Hg (II B group) have been placed in different groups.
(ii) Some elements with higher atomic weights are placed before the elements with lower atomic weights in order to maintain the similar chemical nature of elements. For example,
![]()
(iii) Isotopes did not find any place in the Periodic Table. However, according to Mendeleev’s classification, these should be placed at different places in the Periodic Table.
(All the above three defects were however removed when modern periodic law based on atomic number was given.)
(iv) Position of hydrogen in the Periodic Table is not fixed but is
controversial. ,
(v) Position of elements of group VIII could not be made clear which have been arranged in three triads without any justification.
(vi) It could not explain the even and odd series in IV, V and VI long periods.
(vii) Lanthanides and actinides which were discovered later on, have not been given proper positions in the main frame of Periodic Table.
Question 2.
p-block elements form acidic, basic and amphoteric oxides. Explain each property by giving two examples and also write the reactions of these oxides with water.
Answer:
In p-block, when we move from left to right in a period, the acidic character of the oxides increases due to increase in electronegativity, e.g.,
(i) 2nd period
B2O3 < CO2 < N2O3 acidic character increases.
(ii) 3rd period
Al2O3 < SiO2 < P4O10 < SO3 < Cl2O7 acidic character increases.
On moving down the group, acidic character decreases and basic character increases, e.g.,
![]()
(a) Nature of oxides of 13 group elements
![]()
(b) Nature of oxides of 15 group elements
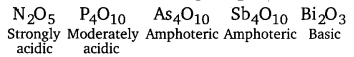
Among the oxides of same element, higher the oxidation state of the element, stronger is the acid. e.g., SO3 is a stronger acid than SO2. B2O3 is weakly acidic and on dissolution in water, it forms orthoboric acid. Orthoboric acid does not act as a protonic acid (it does not ionise) but acts as a weak Lewis acid.
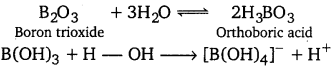
Al2O3 is amphoteric in nature. It is insoluble in water but dissolves in alkalies and react with acids.
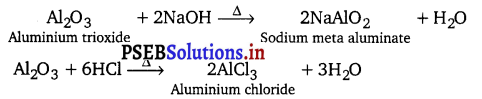
Tl2O is as basic as NaOH due to its lower oxidation state (+1).
Tl2O + 2HCl → 2TlCl + H2O
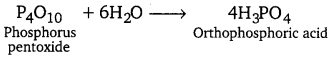
P4O10 on reaction with water gives orthophosporic acid.
Cl2O7 is strongly acidic in nature and on dissolution in water, it gives perchloric acid.